.png)
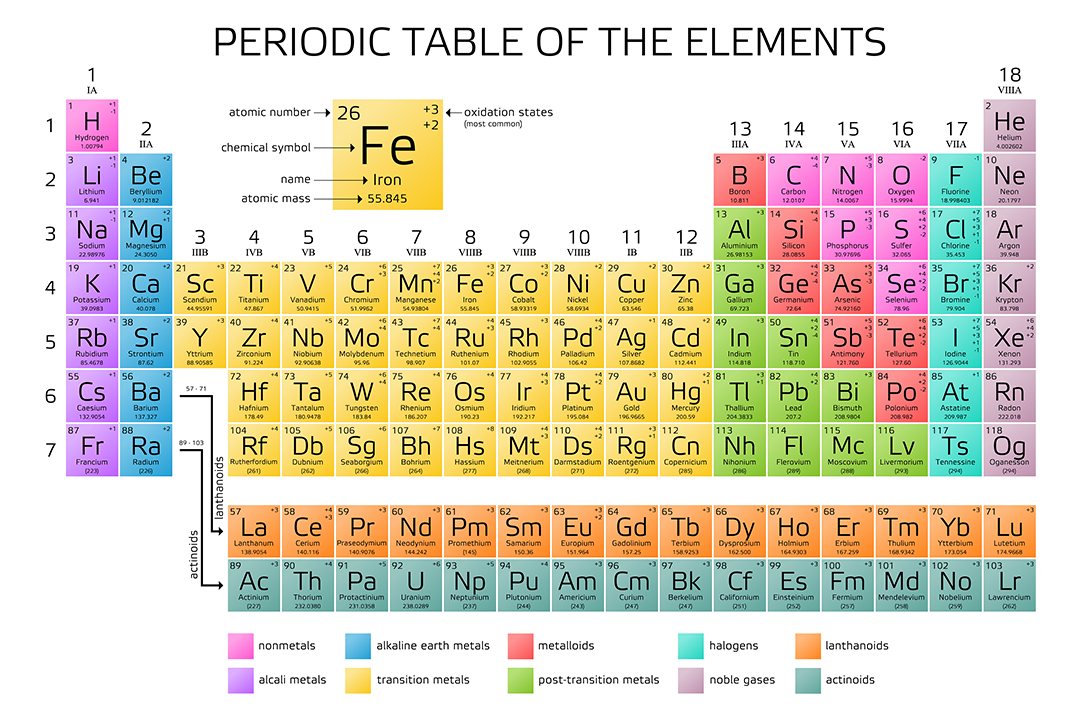
Lanthanoids and Actinoids are numbered as 101 and 102 to separate them in sorting by group. Group: There are only 18 groups in the periodic table that constitute the columns of the table.Elemental compositions of crustal rocks differ between different localities ( see article). Earth crust composition average values are from a report by F.In a sorted list, these elements are shown before other elements that have boiling points >0☌. The density of elements with boiling points below 0☌ is given in g/l.List of Periodic Table elements sorted by → Atomic number No. E.g., sodium ion is represented as Na +1. This number is written with the +/- sign. present at the top right corner (right superscript) of an atomic symbol represents the charge on the atom of that element when it is in its ionic form. present at the top right corner of an atomic symbol represent what?Ī no. For elements whose names are starting with the same letter, the element discovered later will get a two-lettered symbol while the element discovered earlier will get a single-lettered symbol. No two elements can take a similar symbol. of an element is presented in form of a small number at the lower left corner (left subscript) of the symbol of that element. of an element with its symbols?Ītomic no. 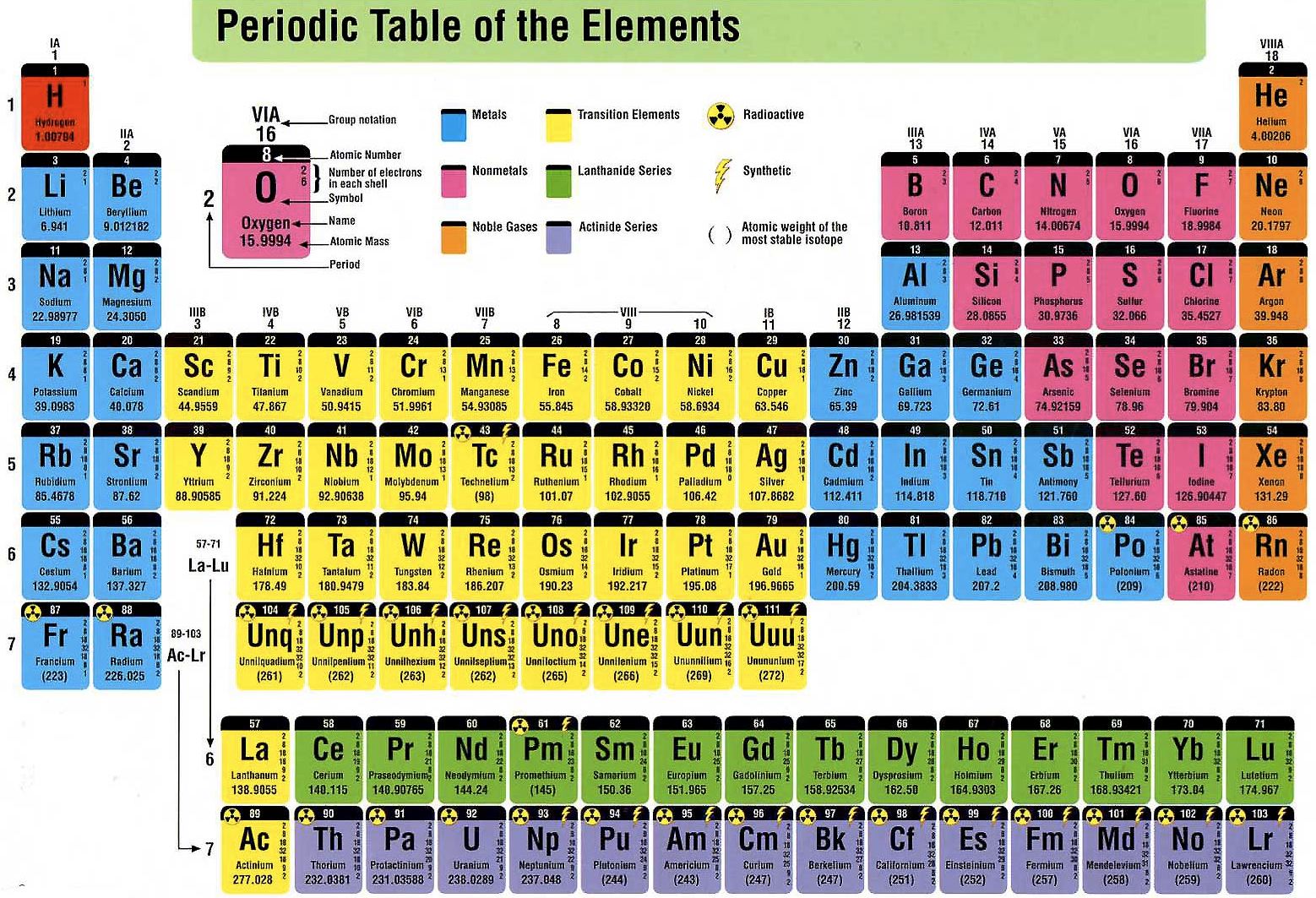
A standard atomic notation shows the symbol, atomic number, mass number and charge (in case of an ion) of the element simultaneously. In standard atomic notation, the name of an element is presented in the form of a symbol with certain super- and sub-scripts. Frequently Asked Questions What is standard atomic notation? The atomic symbols are hence the easiest tools to quickly obtain the essential information about an element. Read more about atomic structures and isotopes While calculating the number of electrons in an ion, we should subtract the positive charge (for cation) from the number of protons and add the negative charge (for anion) to the number of protons. Carbon – 14, on the other hand, has 8 neutrons.īoth isotopes of chlorine contain an equal number of protons and electrons. The carbon-12 has 6 protons, six neutrons, and 6 electrons since the number of protons & electrons must be equal for it to be neutral. 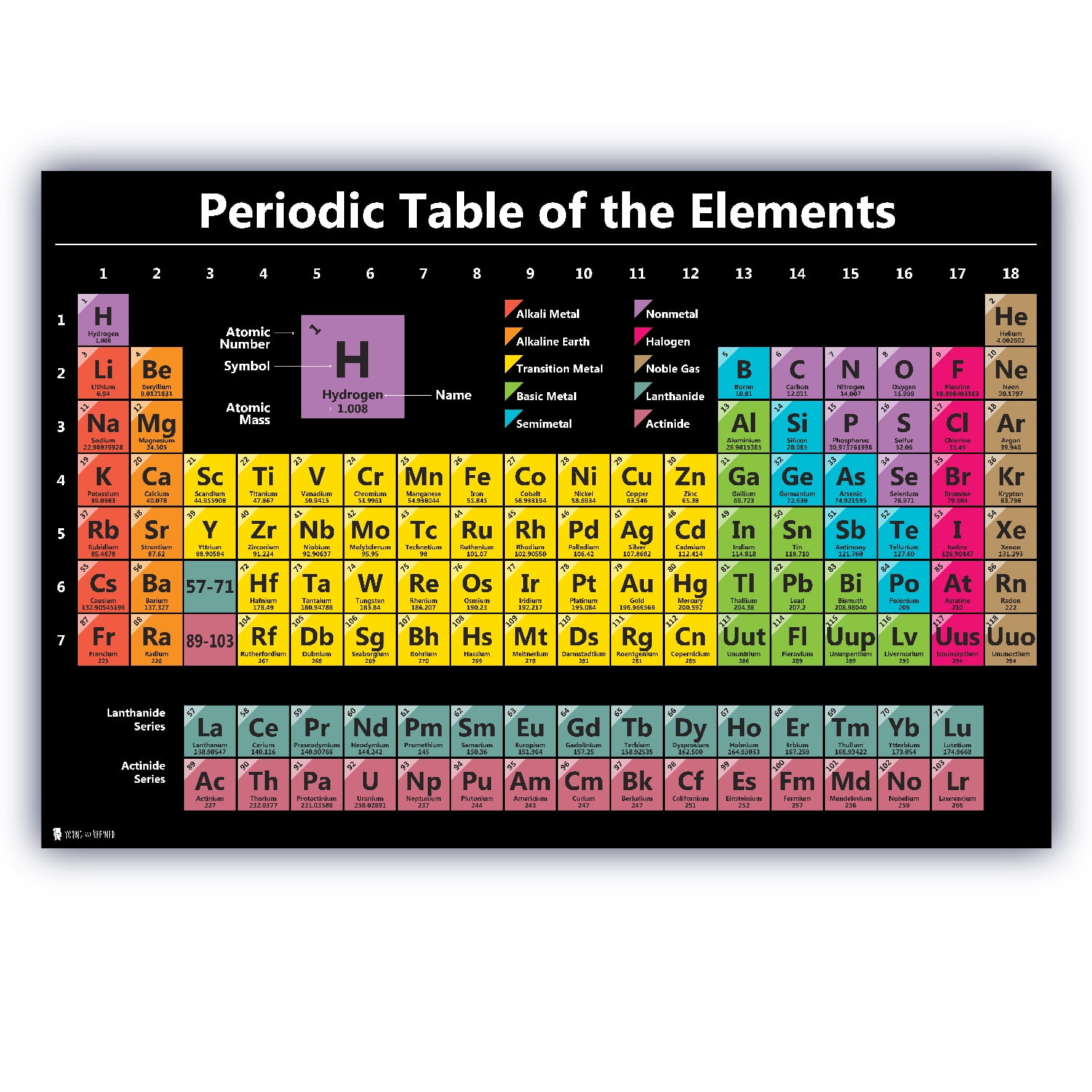
The number of protons, neutrons, and electrons can be easily obtained by the following method.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
